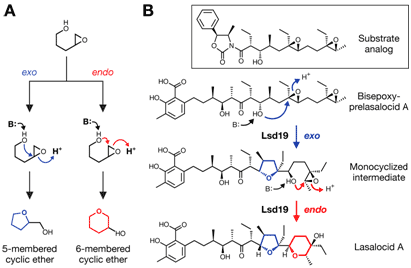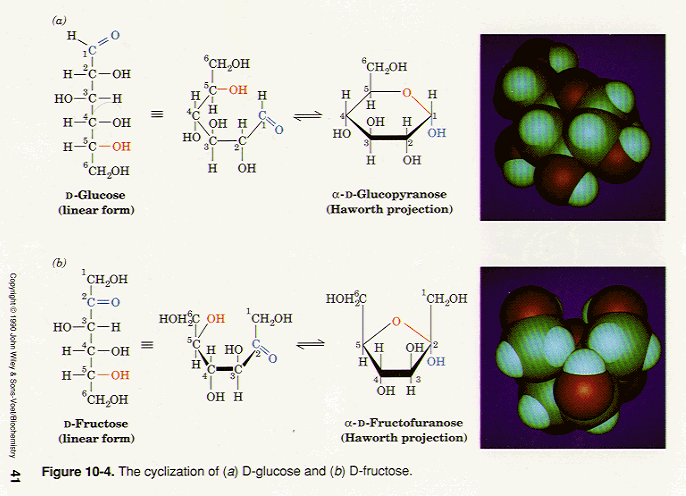
Please explain how this ring closure works - why aren't the "OH" in the closed ring up and down? : r/Mcat
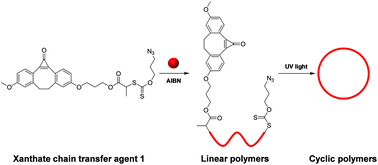
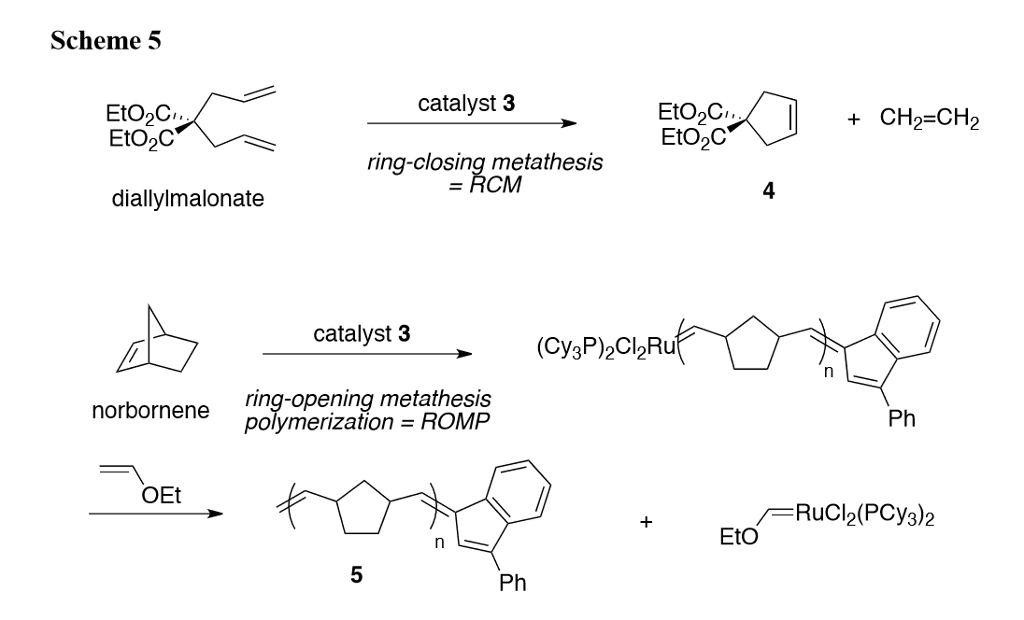
A ring-closure method for preparing cyclic polymers from unconjugated vinyl monomers - Polymer Chemistry (RSC Publishing)
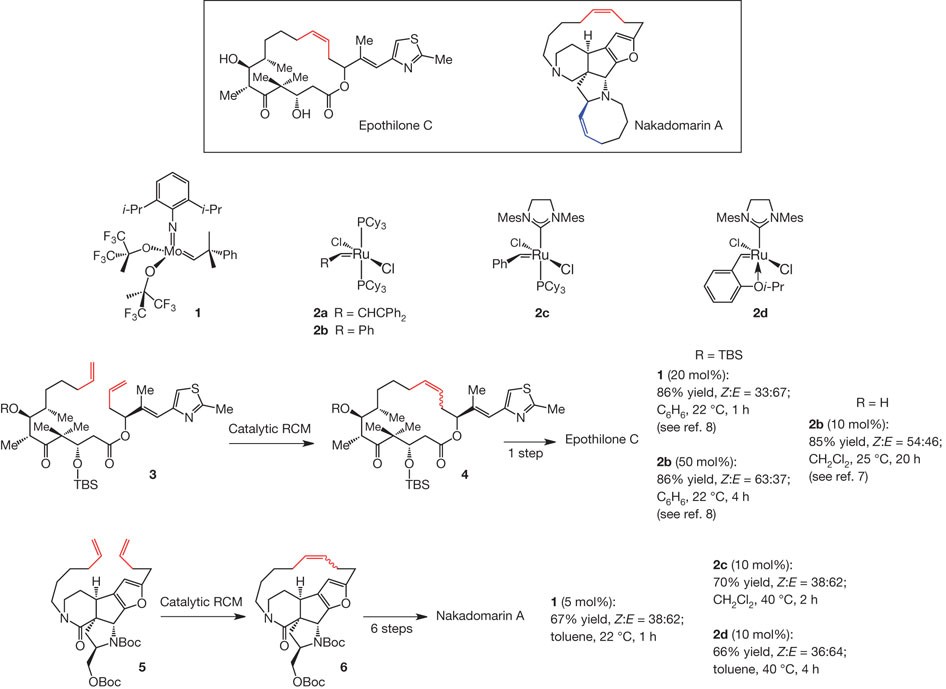
Synthesis of macrocyclic natural products by catalyst-controlled stereoselective ring-closing metathesis | Nature

Michael–Michael Ring-Closure Reactions for a Dihapto-Coordinated Naphthalene Complex of Molybdenum | Organometallics

FeCl3‐Catalyzed Ring‐Closing Carbonyl–Olefin Metathesis - Ma - 2016 - Angewandte Chemie International Edition - Wiley Online Library

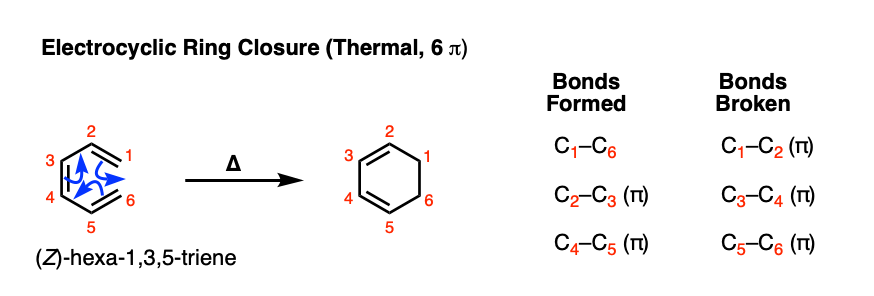
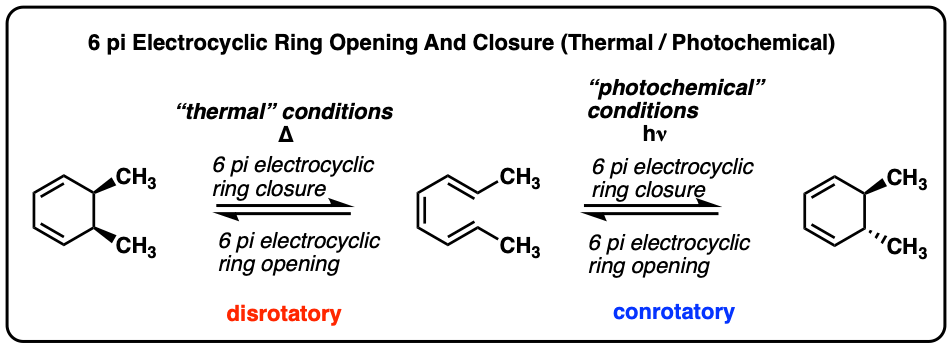
The bicyclic alkene P can be prepared by thermal electrocyclic ring closure from Cyclodecadiene Q or by photochemical electrocyclic ring closure from Cyclodecadiene R. Draw the structures of Q and R, and

Ring-Closing Metathesis of Allylsilanes/Electrophilic Desilylation To Prepare exo-Methylidenecycloalkanes. Short Syntheses of Teucladiol and Poitediol | Journal of the American Chemical Society
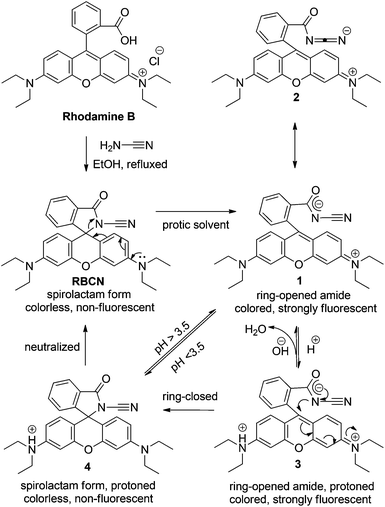
An acid catalyzed reversible ring-opening/ring-closure reaction involving a cyano-rhodamine spirolactam - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C3OB27356C